Osteochondrosis is a common disease in young horse. Before the start of work, it is, in most cases, without associated symptoms. In work, symptoms can show as varying degrees of lameness most often accompanied by swellings: Surgery is sometimes necessary in order to resume sporting activity. In addition to the significant cost of operations, osteochondrosis can lead to drops in performance in sport horses and racehorses, and even compromise their sporting career1-5.
This disease is multifactorial : genetics, feeding of the pregnant mare and the growing foal, the foals’ living conditions, exercise, potential injuries…. all are able to have an influence on it’s development.
Osteochondrosis is therefore not the result of one single cause. It is important to understand osteoarticular development in order to have a better understanding of how and why this condition develops.
This article aims to present osteoarticular development, the biological origins of osteochondrosis as well as risk factors in the gestating mare and growing foal. This is in order to understand that it is possible to help prevent the development of this disease by managing the environment in which the foal grows and develops, that’s to say, right from pregnancy.
SUMMARY
I. The horse’s osteoarticular development
- The growth of long bones
- Organisation of cartilage during horse’s growth
- Is this a common disorder?
- What are the predisposing factors in the development of osteochondrosis?
III. The pregnant mare’s lifestyle
- Mineral balance
- Metabolism of sugars and starch
- Colostrum
- The foal’s living conditions up to the age of 18 months
- Nutrition and growth of the foal before 18 months of age
I. The horse’s osteoarticular development
The growth of long bones
There exists a number of different types of bone in the organism: long bones (for example the femur), flat bones (for example the skull), short bones (bones present in joints) and irregular bones (such as vertebrae, the pelvis). We are going to talk about the growth of long bones as it is their extremities who find themselves within articulations joints affected by osteochondrosis. Bone growth takes place these extremities.
A long bone is made up of :
- A diaphysis, situated at the centre of the bone.
- Two epiphyses, that make up the extremities of the bone and which are covered by articular cartilage. It should be noted that bone is vascularised, cartilage isn’t.
- Two metaphysis located between the diaphysis and the epiphysis, and which are the seat of growth.
Before birth long bones in the foal are composed uniquely of cartilage. As gestation progresses the centre part of this cartilage calcifies, then ossifies : this is the primary ossification centre. In the metaphysis, two Secondary ossification centres appear next: the centre is composed of bone and surrounded by a calcified matrix with cartilage (called growth cartilage) all around. At the base of each secondary ossification centre is the « “growth (or “epiphyseal”) plate ».
As growth progresses, cartilage is replaced by bone, pushing ever further the growth plates. The speed of replacement by bone is faster than that at which cartilage grows, so over time the growth plates become thinner.
In the end, all the cartilage in the growth plates will disappear, this is the closure of the growth plates and thus, the end of growth..
Organisation of cartilage during horse’s growth
Cells making up cartilage are called chondrocytes.
The epiphyseal plate is organised into four different zones :
- A resting zone, in which the chondrocytes are inactive.
- A proliferating zone, where chondrocytes divide rapidly, forming columns.
- A hypertrophic zone, in which the chondrocytes, now much larger, secrete substances that allow the cartilage to calcify, serving as a matrix for bone cells to form bone.
- A calcification zone, where the chondrocytes die, leaving gaps that allow blood vessels and bone cells (osteocytes) to penetrate into the tissue.
II. What is osteochondrosis?
Osteochondrosis is a development disorder that evolves in the foal up to the age of 18 months9. Lesions may appear and naturally resorb themselves within this time frame. The disorder is defined by a disruption in the maturation of the cartilage involved in bone growth which may lead to the releasing of fragments into the joints7.
This disorder is not associated with osteoarthritis and is not of inflammatory origin.
Generally, osteochondrosis results from a lack of vascularisation in the growth cartilage situated at the cartilage/ bone junction. In this case, cartilage persists as bone cannot form to replace it. Thus, the junction zone between the cartilage and the bone becomes irregular and the cartilage becomes soft and fragile8-15.
The reason behind this lack of vascularisation has not yet been totally solved. A number of theories have been put forward over many years. Today, the validated hypothesis seems to be the one of the persistence of chondrocytes in the hypertrophic zone and thus the cartilage within the subchondral bone16. Furthermore, within lesions, chondrocytes show a break in the balance between deterioration/ maintenance factors of the cartilage matrix and vascularisation factors17.
Thus the cartilage matrix cannot be replaced by the bone matrix, and will not have enough factors to attract blood vessels.
The disorder divides into three stages :
- 1. Osteochondrosis latens: There are very small zones without blood vessels in the growth plate, these lesions may regress, even disappear, they are not visible on x-rays,.
- 2. Osteochondrosis manifesta: cartilage is no longer replaced by bone, it becomes soft and fragile. This can lead to the formation of cracks, zones with a loss of density, or irregularities on the cartilage surface which are visible on x-rays.
- 3. Osteochondrosis dissecans (OCD) or cyst : The split/ crack is widespread and/or cartilage fragments are released into the joint. Osteochondrosis manifesta can also lead to the forming of a bone cyst, that is to say a liquid filled hole in the bone7.
If you have known about this disorder for a certain time, you will have heard of “osteochondritis dissecans”. In medicine, the term “itis” means a disease of inflammatory origin. As this is not the case this name was modified a few years back to osteochondrosis and osteochondritis is no longer used today. .
Is this a common disorder?
The prevalence of osteochondrosis depends on the breed and the country studied.
Indeed, osteochondrosis is more frequent seen in fast growing breeds such as thoroughbreds, trotters (Standardbreds), riding horses… than slow growing breeds such as Icelandic horses, ponies and draft horses.
These differences between breeds can be explained by the fact that in horses a larger number of blood vessels must be incorporated into the cartilage during growth than in ponies. For example, in Trotters, growth cartilage is thicker, with a larger number of blood vessels than in the Fjord333. Therefore there is a higher risk of vascularisation problems and thus of developing the disorder15. Genetics is an important development factor in this condition, all the more since it is known to exist within breeds, and certain lines being more affected than others.
It must however be noted thatheredity varies between breeds and the joints affected, and is not the only cause of lesions developing. Environmental factors, right from the start of foetal life, can modify the development of this disorder that is programmed by the genetic patrimony of the foal.
What are the predisposing factors in the development of osteochondrosis?
The environmental factors predisposing to the development of osteochondrosis can be divided into three categories:
- Injuries and biomechanical constraints,
- The feeding and metabolism of the mare and foal,
- The foal’s living conditions.
The hypothesis of mechanical demands is substantiated by the fact that certain sites in certain joints are more affected than others. Thus, constraints of weight, of pressure, of friction… would be greater at these locations and play a role in the development of the disease by weakening the joint12.
The pregnant mare’s lifestyle
Osteochondral lesions have been observed in foals just 2 days old10,15,34,35. At this age, the foal has not yet spent long enough outside of his dam for the postnatal environment to be the explication for the appearance of lesions. Furthermore, during the final two-thirds of gestation, growth cartilage in the foal undergoes important vascular changes36, thus this is a particularly critical period in the growth and development of bone.
Mineral balance
Copper is an essential element in osteoarticular development..
Supplementing pregnant mares with copper above their requirement levels does not always reduce the incidence of osteochondrosis in foals37–40. However, adequate supplementation in copper during the last three months of gestation increases the hepatic stock of copper in the new-born foal38,39. Mare’s milk being naturally deficient in copper, the foal must be born with a big enough hepatic stock to meet his requirements for growth.
Supplementing milking mares with copper does not increase the amount of copper in the milk41. It is therefore the mineral balance of the ration and the absence of deficiencies in pregnant mares that seems to have a significant influence over the development of osteochondral lesions in the foal.
Metabolism of sugars and starch
Glucose is a very important source of energy for the organism.Thus the body must have a certain quantity of glucose at it’s disposal in order to function. However, in too large a quantity glucose is toxic. Thus the blood sugar level (glycaemia) must be regulated. When the glycaemia increases (after a feed for example), the pancreas produces insulin whose role is to stimulate the storage of glucose in muscle and fatty tissues, thus enabling the glycaemia to return to a normal level..
The role played by insulinn in chondrocyte growth and bone development is still under discussion. Insulin is considered as a growth factor, notably known for stimulating the multiplication of chondrocytes33, but also with the ability of modifying the expression of a number of genes. It can in this way, when too much is present, disturb the balance between cartilage persisting and deteriorating and thus bone growth.
Distributing feeds rich in starch to pregnant mares increases the risk of their foals developing osteochondrosis.. This risk is even greater if starch in cereals is predigested, having undergone a technological treatment beforehand such as such as flaking, micronising, puffing and extrusion. Furthermore, soluble sugars (such as those inmolasses) lead to an even steeper rise in the glycaemia and blood insulin level after the feed. An increase in insulin production after a feed in the pregnant mare is therefore related to an increase in the number of foals showing osteochondral lesions30,42,43.
Foetal chondrocytes are particularly sensitive to insulin, thus the time the foal is in utero should not be neglected44. Furthermore, mares ingesting more than 200g of starch per 100 kg live-weight per day (and so more than 100g/100kg live-weight per feed) produce more foals suffering from osteochondrosis than mares ingesting less than 200g of starch per 100 kg live-weight and per day (and thus less than100g/100kg live-weight per feed) (pers. comm., Robles et al.,).
The development of resistance to insulin can be caused by the long term consumption of cereals with a high glycemic index and /or disorders such as obesity in the mare. Insulin resistance is notably characterized by a drop in the ability of insulin to stimulate the storing of glucose. Mares who suffer from resistance to insulin, because of obesity or through being fed large amounts of starch during gestation, also produce a greater number of foals suffering from osteochondral lesions42,45.
Thus the metabolism of glucose in the gestating mare is an important factor, and its disturbance, as a result of feeding, can alter bone development in foals.
⏩ Also read : Preventing osteoarticular disorders
Colostrum
Finally, mares producing lower quality colostrum at birth, that is to say with lower level of anti-bodies (immunoglobulins G), have higher numbers of foals suffering from osteochondral lesions18,43. Colostrum quality can be altered by feeding during gestation, and notably by an excessively rich diet with a too high a glycemic index18,46,47. The foal is born with limited immune defences. The dam must be able, via milk, to transfer anti-bodies to give the foal protection until his own immune defence system is established.
The relationship between poor quality colostrum and osteochondrosis has yet to be explained.
The foal’s lifestyle
The foal’s living conditions up to the age of 18 months
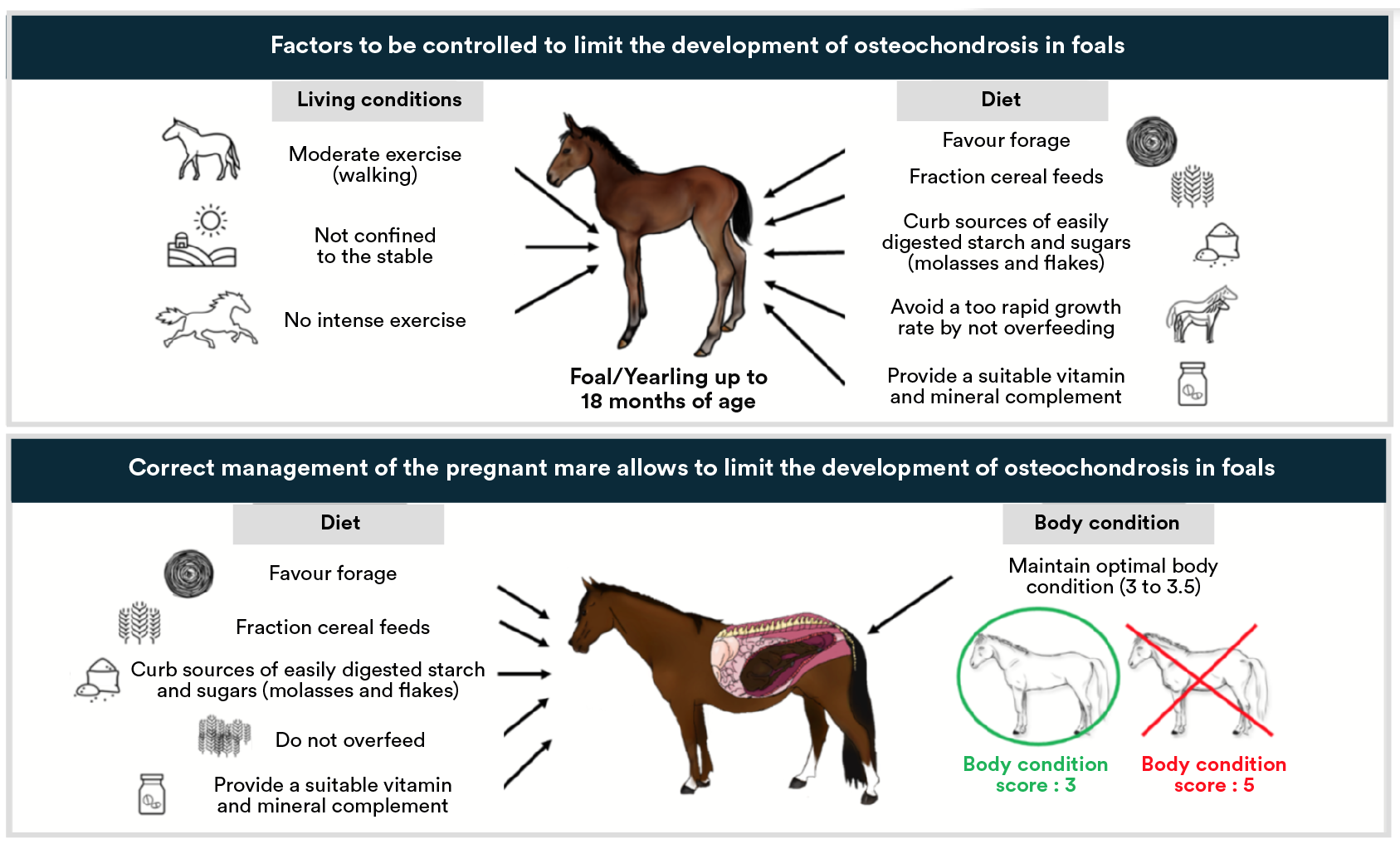
Foals living out at grass 24/7 are at less risk of developing osteochondrosis than foals out at grass during the day and stabled at night. This is related to the sudden and very large increase in physical activity in foals stabled overnight and thus to an increase in biomechanical constraints and the risk of injuries47.
However, this has not been observed in every epidemiological study undertaken and should be nuanced in relation to the manner in which foals are turned out, the size of paddock and even herd size28. In addition, sustained and controlled exercise (pre-training…) during growth is an aggravating factor for existing lesions, all the more so when the osteoarticular system of the youngster isn’t ready for hard work.
In any event, foals who are able to exercise moderately (walk) on a regular basis are less at risk of developing osteochondral lesions than foals confined to the stable48,50.Moreover, cartilage maturation is of particular importance during the first 5 months of the foal’s life and may be retarded if the foal remains stabled, and thus can also lead to changes in the development of joints51.
Nutrition and growth of the foal before 18 months of age
One of the most important factors in bone growth is the balanced provision of minerals. Bones are composed of a protein based matrix, which gives them elastic properties (this allows bones to have a certain ability to deform before breaking), and crystals that are rich in calcium and phosphorus. Certain minerals and trace elements such as copper are also necessary to create elements that make up the protein based matrix.
Hence, copper deficiency in the growing foal is associated with an increase in osteoarticular lesions52-54. In addition, an excess of phosphorus in relation to calcium in the diets of foals aged between 2 and 6 months old would seem to be related to an increase in the development of osteochondral lesions56.
Calcium absorption enters into competition with that of phosphorus, a diet too rich in phosphorus thus leads to poor absorption of calcium by foals who are unable to use it for bone growth. However, a diet containing excess calcium will not be prejudicial to the osteoarticular development of the foal.
Foals that grow too quickly and who are overfed have a greater risk of developing osteochondral lesions22,23,55–57. Indeed, overfeeding leads to an altered development of cartilage and thus to poor bone growth in the joints58. Maximum growth does not signify optimal growth as bone cannot simultaneously grow and mineralise correctly.
Not only is the amount of energy of importance, but also its quality : Foals fed with cereals have a greater risk of developing osteochondral lesions and have less chance of recovering from these lesions than those exclusively fed on forage48. This relationship becomes all the more evident when cereals fed have a high glycemic index, thus sharply increasing the glycaemia and blood insulin levels in foals after feeds59,60. We sometimes hear that the amount of protein fed to foals is a factor to take into consideration in the development of osteochondrosis.
If few research projects have studied this subject, one of them has demonstrated that it is an excess of energy and not an excess of protein that is at the origin of the development of this pathology58.
Conclusion
To conclude, osteochondrosis is a disorder with a strong genetic component but is equally heavily dependant on the environment in which the foal finds himself from his moment of conception. Careful management of the pregnant mare and growing foal up to the age of 18 months thus will determine the osteoarticular health of the foal. It should be known that the early environment in which the foal develops will also programme other aspects of his health at an adult age, such as his growth, his adiposity, his metabolizing of carbohydrates (a role of capital importance in sporting performance), muscular capacity, reproductive capability, or even his behaviour.
Therefore, good and careful management of pregnant mares and growing foals helps the genetic potential of the foal to be reached and puts luck on his side.
Practical advice for broodmares and foals aimed at preventing osteochondrosis :
- Favour the use of forage,
- » Restrict the amount of sugar and digestible starch in the diet : feed a concentrate ration based on the use of unflaked, unmicronised, non-puffed or non-extruded barley starch, and without added sugar (molasses for example),
- Carefully think about the amount of starch fed daily and split the concentrate feed into as many meals possible,
- » Use a suitable vitamin and mineral supplement,
- Do not overfeed, in order to maintain an optimal condition and growth rate.
In regards to quantities of starch, for a gestating mare we recommend not exceeding 100g of starch per 100kg live-weight per meal. For foals, the maximum quantity of starch not to be exceeded per meal has yet to be determined.
Bibliography
1. Jørgensen, H., Proschowskyt, H., Nne, J., Willebergq, P. & Hesselholt, M. The significance of routine radiographic findings with respect to subsequent racing performance and longevity in Standard bred trotters. Equine Vet. J. 29, 55–59 (1997).
2. Laws, E., Richardson, D., Ross, M. & Moyer, W. Racing performance of Standardbreds after conservative and surgical treatment for tarsocrural osteochondrosis. Equine Vet. J. 25, 199–202 (1993).
3. Robert, C., Valette, J. & Denoix, J. Correlation between routine radiographic findings and early racing career in French Trotters. Equine Vet. J. 36, 473–478 (2006).
4. Kane, A. et al. Radiographic changes in Thoroughbred yearlings . Part 2 : Associations with racing performance. Equine Vet. J. 35, 366–374 (2003).
5. Verwilghen, D. et al. Do developmental orthopaedic disorders influence future jumping performances in Warmblood stallions ? Equine Vet. J. 45, 578–581 (2013).
6. Mackie, E., Tatarczuch, L. & Mirams, M. The skeleton: A multi-functional complex organ. The growth plate chondrocyte and endochondral ossification. J. Endocrinol. 211, 109–121 (2011).
7. Van Weeren, P. Etiology, Diagnosis, and Treatment of OC(D). Clin. Tech. Equine Pract. 5, 248–258 (2006).
8. Ytrehus, B., Carlson, C. & Ekman, S. Etiology and pathogenesis of osteochondrosis. Vet. Pathol. 44, 429–448 (2007).
9. Jacquet, S., Robert, C., Valette, J. & Denoix, J. Evolution of radiological findings detected in the limbs of 321 young horses between the ages of 6 and 18 months. Vet. J. 197, 58–64 (2013).
10. Carlson, C., Cullins, L. & Meuten, D. Osteochondrosis of the articular-epiphyseal cartilage complex in young horses: evidence for a defect in cartilage canal blood supply. Veterinary Pathology 32 641-647 (1995).
11. Jeffcott, L. & Henson, F. Studies on growth cartilage in the horse and their application to aetiopathogenesis of dyschondroplasia (osteochondrosis). Vet. J. 156, 177–192 (1998).
12. Douglas, J. Pathogenesis of osteochondrosis. in Diagnosis and management of lameness in the horse (eds. Ross, M. & Dyson, S.) 617–625 (Saunders, Elsevier, 2011).
13. Kinsley, M., Semevolos, S. & DuesterdieckZellmer, K. Wnt/β-catenin signaling of cartilage canal and osteochondral junction chondrocytes and full thickness cartilage in early equine osteochondrosis. J. Orthop. Res. 33, 1433–1438 (2015).
14. Olstad, K., Ekman, S. & Carlson, C. An Update on the pathogenesis of Osteochondrosis. Vet. Pathol. 52, 785–802 (2015).
15. Mirams, M. et al. Altered gene expression in Early Osteochondrosis lesions. J. Orthop. Res. 27, 452–457 (2009).
16. Mirams, M. et al. Identification of novel osteochondrosis - Associated genes. J. Orthop. Res. 34, 404–411 (2016).
17. Hendrickson, E. et al. Comparison of the blood supply to the articular-epiphyseal growth complex in horse vs . pony foals. Equine Vet. J. 47, 326–332 (2015).
18. Robles, M. Influence du métabolisme maternel sur la fonction placentaire et la santé du poulain. (Thèse de doctorat, Université Paris-Saclay - AgroParisTech, 2017).
19. Björnsdóttir, S., Axelsson, M., Eksell, P., Sigurdsson, H. & Carlsten, J. Radiographic and clinical survey of degenerative joint disease in the distal tarsal joints in Icelandic horses. Equine Vet. J. 32, 268–272 (2000).
20. Strand, E., Braathen, L., Hellsten, M., Huse-olsen, L. & Björnsdóttir, S. Radiographic closure time of appendicular growth plates in the Icelandic horse. Acta Vet. Scand. 49, 19 (2007).
21. Riley, C. et al. Osteochondritis dessicans and subchondral cystic lesions in draft horses: A retrospective study. Can. Vet. J. 39, 627–633 (1998).
22. Pagan, J. The incidence of developmental orthopedic disease (DOD) on a Kentucky Thoroughbred farm. Adv. Equine Nutr. 469– 475 (1998).
23. Lepeule, J. et al. Association of growth, feeding practices and exercise conditions with the prevalence of Developmental Orthopaedic Disease in limbs of French foals at weaning. Prev. Vet. Med. 89, 167–177 (2009).
24. Combs, T. et al. A Transgenic Mouse with a Deletion in the Collagenous Domain of Adiponectin Displays Elevated Circulating Adiponectin and Improved Insulin Sensitivity. Endocrinology 145, 367– 383 (2004).
25. Bastos, L. et al. Incidence of Juvenile Osteochondral conditions in thoroughbred weanlings in the south of Brazil. J. Equine Vet. Sci. (2017). doi:10.1016/j.jevs.2017.02.008
26. Grøndahl, A. The incidence of osteochondrosis in the tibiotarsal joint of Norwegian Standardbred trotters. J. EquineVet. Sci. 11, 272–274 (1991).
27. Sandgren, B. Bony fragments in the tarsocrural and metacarpo- or metatarsophalangeal joints in the Standardbred horse - a radiographic survey. Equine Vet. J. 6, 66–70 (1987).
28. Lykkjen, S., Roed, K. & Dolvik, N. Osteochondrosis and osteochondral fragments in Standardbred trotters: Prevalence and relationships. Equine Vet. J. 44, 332–338 (2011).
29. Vander Heyden, L. et al. Prévalence de l’ostéochondrose chez le cheval de sport en Wallonie. Ann. Médecine Vétérinaire 152, 61–70 (2008).
30. Vander Heyden, L. et al. Association of breeding conditions with prevalence of osteochondrosis in foals. Vet. Rec. 172, 68 (2012).
31. Vos, N. et al. Incidence of osteochondrosis (dissecans) in Dutch warmblood horses presented for pre-purchase examination. Ir. Vet. J. 61, 33–37 (2008).
32. Van Grevenhof, E. et al. Prevalence of various radiographic manifestations of osteochondrosis and their correlations between and within joints in Dutch Warmblood horses. Equine Vet. J. 41, 11–16 (2009).
33. Jönsson, L., Dalin, G., Egenvall, A., Roepstorff, L. & Philipsson, J. Equine hospital data as a source for study of prevalence and heritability of osteochondrosis and palmar / plantar osseous fragments of Swedish Warmblood horses. Equine Vet. J. 43, 695– 700 (2011).
34. Henson, F., Davies, M. & Jeffcott, L. Equine dyschondroplasia (osteochondrosis)-histological findings and type VI collagen localization. Vet. J. 154, 53–62 (1997).
35. Rejnö, S. & Strömberg, B. Osteochondrosis in the horse. II. Pathology. Acta Radiol. Suppl. 358, 153–78 (1978).
36. Lecocq, M. et al. Cartilage matrix changes in the developing epiphysis : Early events on the pathway to equine osteochondrosis ? Equine Vet. J. 40, 442– 454 (2008).
37. Knight, D. et al. The effects of copper supplementation on the prevalence of cartilage lesions in foals. Equine Vet. J. 22, 426–432 (1990).
38. Pearce, S., Grace, N., Wichtel, J., Firth, E. & Fennessy, P. Effect of copper supplementation on copper status of pregnant mares and foals. Equine Vet. J. 30, 200–203 (1998).
39. Pearce, S. G., Firth, E. C., Grace, N. D. & Fennessy, P. F. Effect of copper supplementation on the evidence of developmental orthopaedic disease in pasture-fed New Zealand Thoroughbreds. Equine Vet. J. 30, 211–218 (1998).
40. Winkelsett, S., Vervuert, I., Granel, M. & Borchers, A. Feeding practice in Warm bloodmares and foals and the incidence to osteochondrosis. Pferdeheilkunde 21, 124– 126 (2003).
41. Kavazis, A., Kivipelto, J. & Ott, E. Supplementation of broodmares with copper, zinc, iron, manganese, cobalt, iodine, and selenium. J. Equine Vet. Sci. 22, 460–464 (2002).
42. Peugnet, P. et al. Effects of moderate amounts of barley in late pregnancy on growth, glucose metabolism and osteoarticular status of pre- weaning horses. PLoS One 10, e0122596 (2015).
43. Caure, S. & Lebreton, P. Ostéochondrose chez le trotteur au sevrage et corrélation avec divers paramètres. Prat. Vétérinaire Equine 36, 47–57 (2004).
44. Henson, F. et al. Effects of insulin and insulin-like growth factors I and II on the growth of equine fetal and neonatal chondrocytes. Equine Vet. J. 29, 441–447 (1997).
45. Robles, M. et al. Maternal obesity increases insulin resistance, low-grade inflammation and osteochondrosis lesions in foals and yearlings until 18 months of age. PLoS One 13, (2018).
46. Thorson, J. et al. Effect of selenium supplementation and plane of nutrition on mares and their foals: Foaling data. J. Anim. Sci. 88, 982–990 (2010).
47. Coverdale, J., Hammer, C. & Walter, K. Nutritional programming and the impact on mare and foal performance. J. Anim. Sci. 93, 3261–3267 (2015).
48. Mendoza, L. et al. Impact of feeding and housing on the development of osteochondrosis in foals — A longitudinal study. Prev. Vet. Med. 127, 10–14 (2016).
49. van Weeren, P. & Barneveld, A. The effect of exercise on the distribution and manifestation of osteochondrotic lesions in the Warmblood foal. Equine Vet. J. Suppl. 31, 16–25 (1999).
50. Lepeule, J. et al. Association of growth, feeding practices and exercise conditions with the severity of the osteoarticular status of limbs in French foals. Vet. J. 197, 65–71 (2013).
51. Brama, P., TeKoppele, J., Bank, R., Barneveld, A. & van Weeren, P. Development of. biochemical heterogeneity of articular cartilage: influences of age and exercise. Equine Vet. J. 34, 265–269 (2002).
52. Hildebran, S. & Hunt, J. Copperresponsive epiphytis and tendon contracture in a foal. in The Roles of Amino Acid Chelates in Animal Nutrition (ed. Ashmead, H.) 400– 410 (Noyes Publications, 1993).
53. Hurtig, M., Green, S., Dobson, H., Mikuni-Takagaki, Y. & Choi, J. Correlative study of defective cartilage and bone growth in foals fed a low-copper diet. Equine Vet. J. 25, 66–73 (1993).
54. Coskun, A., Ozdemir, O., Erol, M. & Kirbiyik, H. The relationship of copper concentrations in feed and plasma to developmental orthopedic disease in foals. Vet. Arh. 86, 287–294 (2016).
55. Savage, C., McCarthy, R. & Jeffcott, L. Effects of dietary energy and protein induction on dyschondroplasia in foals. Equine Vet. J. Suppl. 16, 74–79 (1993).
56. Savage, C., McCarthy, R. & Jeffcott, L. Effects of dietary phosphorus and calcium on induction of dyschondroplasia in foals. Equine Vet. J. 16, 74–79 (1993).
57. Donabédian, M. et al. Effect of fast vs. moderate growth rate related to nutrient intake on developmental orthopaedic disease in the horse. Anim. Res. 55, 471–486 (2006).
58. Glade, M. & Belling, T. A dietary etiology for osteochondrotic cartilage. J. Equine Vet. Sci. 6, 151–155 (1986).
59. Ralston, S. Hyperglycemia/ hyperinsulinemia after feeding a meal of grain to young horses with osteochondritis dissecans (OCD) lesions. Pferdeheilkunde 12, 320–322 (1996).
60. Pagan, J., Geor, R., Caddel, S., Pryor, P. & Hoekstra, K. The Relationship Between Glycemic Response and the Incidence of OCD in Thoroughbred Weanlings: A Field Study. in Proceedings of the 47th AAEP Annual Convention 47, 322–325 (2001).




